
Insights+: EMA Marketing Authorization of New Drugs in February 2023
Shots:
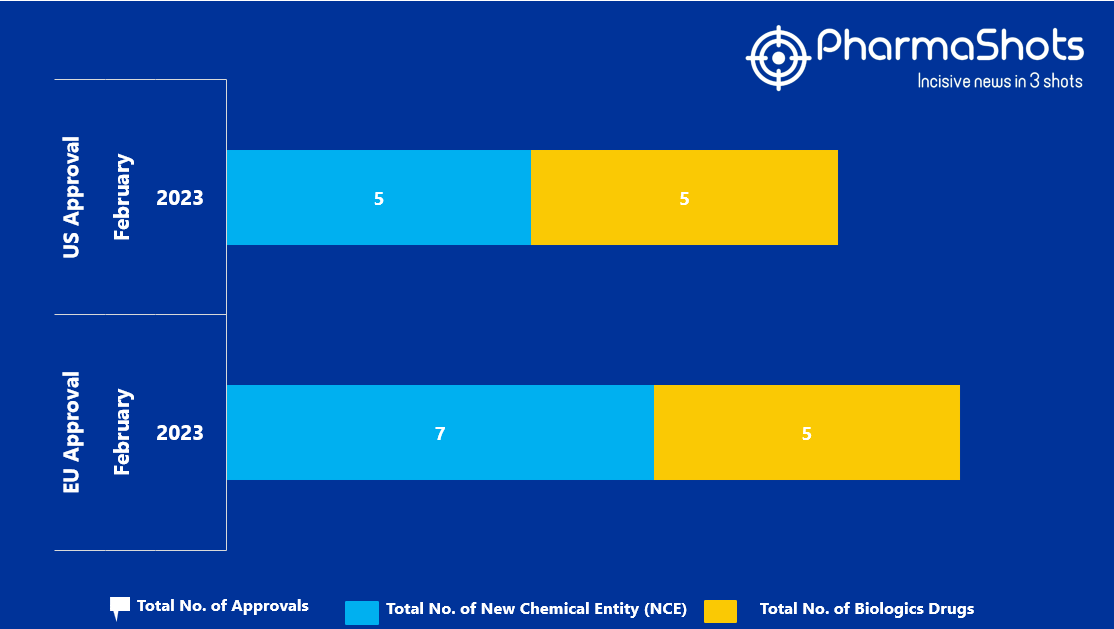
- The EMA approved 7 New Chemical Entity (NCE) and 5 Biologic Drugs in February 2023, leading to treatments for patients and advances in the healthcare industry
- In February 2023, the major highlights drugs were Forxiga’s Approval for symptomatic chronic heart failure, Fintepla for adjunctive treatment of seizures associated with lennox-gastaut syndrome
- PharmaShots has compiled a list of a total of 12 new drugs approved by the EMA in February 2023
Hemlibra
Active ingredient: emicizumab Approved: February 01, 2023
Company: Roche Disease: Haemophilia A
- The EC has approved a label expansion of Hemlibra (bispecific factor IXa- and factor X-directed Ab) in patients with a mod. haemophilia A. The approval was based on the results of the P-III trial (HAVEN 6) evaluating the safety, efficacy, PK & PD of Hemlibra
- The results showed effective bleed control & favorable safety profile, 70.8% had mod. haemophilia A without factor VIII inhibitors with no new safety signals, patients experienced no bleeds/no spontaneous bleeds/no joint bleeds were 66.7%/81.9%/88.9%, respectively
- The decision was also based on real-world data while the label expansion will provide an effective & convenient prophylactic treatment option for patients with a mod. haemophilia A with a sev. bleeding phenotype
Forxiga
Active ingredient: dapagliflozin Approved: February 07, 2023
Company: AstraZeneca Disease: Heart Failure
- The approval was based on the P-III trial (DELIVER) evaluating Forxiga vs PBO in 6263 HF patients with LVEF ≥40% with/out T2D. The primary composite EPs were the time to the first occurrence of CV death, hHF, or an urgent HF visit. & 2EPs incl. the total no. of HF events & CV death, change from baseline in the total symptom score of KCCQ @8mos., time to the occurrence of CV death & death from any cause
- Forxiga was found to be 1st HF drug to show a mortality benefit across the full ejection fraction range in the prespecified pooled analysis of the P-III (DELIVER) & (DAPA-HF) trials
- Forxiga was approved in 100+ countries globally for T2D, HFrEF & CKD incl. the US, the EU, China & Japan. The therapy has received regulatory approvals in Great Britain, Japan & Turkey
Hemgenix
Active ingredient: etranacogene dezaparvovec Approved: February 21, 2023
Company: CSL Disease: Hemophilia B
- The EC has granted CMA for Hemgenix in adults with sev. & moderately sev. hemophilia B without a history of Factor IX inhibitors
- The approval was based on the P-III trial (HOPE-B) results evaluating Hemgenix in 54 adult patients. The results showed a stable & durable increase in mean Factor IX activity levels with a mean Factor IX activity of 36.9%, leading to an ABR reduction of 64%, 96% discontinued routine Factor IX prophylaxis & 97% reduction in mean Factor IX consumption @18mos. post-treatment, was well-tolerated with no serious TRAEs
- The EC’s decision will be valid in all EU member states, incl. EEA countries of Iceland, Norway & Liechtenstein. Hemgenix’s submission is currently under MHRA review in the UK
Imfinzi
Active ingredient: durvalumab Approved: February 22, 2023
Company: AstraZeneca Disease: Liver and Non-Small Cell Lung Cancer
- The approval was based on the P-III trial (HIMALAYA) evaluating Imjudo (300mg) + Imfinzi (1500mg, q4w) vs sorafenib in 1324 patients with HCC & P-III (POSEIDON) trial of Imfinzi + Imjudo & CT vs CT alone in 1013 patients with metastatic NSCLC
- In (HIMALAYA) & (POSEIDON) trials published in the NEJM & Journal of Clinical Oncology, 22% & 23% reduction in risk of death, m-OS (16.4 vs 13.8mos.) & (14.0 vs 11.7mos.), patients were alive (31% vs 20%) @3yrs. & (33% vs 22%) @2yrs. & no new safety signals were seen
- In (POSEIDON) trial, 28% reduction in risk of disease progression or death, m-PFS (6.2 vs 4.8mos.) & updated results @4yrs. of follow-up showed a sustained survival benefit, 25% reduction in risk of death, m-OS (14 vs 11.7mos.), 25% vs 13.6% were alive @3yr.
Triumeq PD
Active ingredient: dolutegravir, abacavir and lamivudine Approved: February 22, 2023
Company: ViiV Healthcare Disease: Human Immunodeficiency Virus Type 1
- The company received marketing authorization from the EC for Triumeq PD to treat paediatric patients with HIV-1. The authorization also includes the label extension of Triumeq & lowers the minimum weight of a child who can be prescribed this medicine to 25kgs from 40 kg
- The authorization was based on the US FDA’s approval of Triumeq PD in 2022. In 2021, 52% of children aged ≤14yrs. living with HIV had access to antiretroviral medications with paediatric optimizations.
- Triumeq is a dispersible tablet formulation of the fixed-dose combination of abacavir, dolutegravir, and lamivudine for the treatment of paediatric patients with HIV-1
Akeega
Active ingredient: niraparib Approved: February 24, 2023
Company: Janssen Disease: Castration-Resistant Prostate Cancer
- The opinion was based on the P-III study evaluating niraparib (200mg, qd) + abiraterone acetate & prednisone vs PBO + AA & prednisone in a ratio (1:1) in 765 patients with/out certain HRR gene alterations
- Improvement in rPFS in all HRR+ patients & 47% risk reduction for rPFS in patients with BRCA1/2 gene mutations. In the updated 2nd interim analysis, consistent & significant treatment effect in rPFS at a median follow-up of 24.8mos. in the BRCA subgroup with an m-rPFS (19.5 vs 10.9mos.)
- In the BRCA subgroup, improved OS, time to symptomatic progression & consistent improvement of TCC were reported, grade 3/4 AEs for HRR gene alterations (67% vs 46.4%), and AEs leading to treatment discontinuation (10.8% vs 4.7%) & maintained overall QoL
Vafseo
Active ingredient: vadadustat Approved: February 24, 2023
Company: Akebia Disease: Chronic Kidney Disease
- The EMA’s CHMP has adopted a positive opinion recommending approval of Vafseo (HIF-PH inhibitor) for symptomatic anemia associated with CKD in adults on chronic maintenance dialysis. The EC’s decision is expected in ~2mos.
- The opinion was based on the comprehensive development program incl. the P-III (INNO2VATE) program of vadadustat. The trial met its primary & secondary efficacy EPs in each of 2 (INNO2VATE) studies i.e., (Correction/Conversion and Conversion) & showed non-inferiority to darbepoetin alfa
- The therapy also achieved primary safety EPs i.e., non-inferiority of vadadustat over darbepoetin alfa in time to 1st occurrence of major adverse cardiovascular EPs. The EC’s decision will valid for all 27 EU member states, Iceland, Norway & Liechtenstein
Opzelura
Active ingredient: ruxolitinib Approved: February 24, 2023
Company: Incyte Disease: Non- Segmental Vitiligo
- The EMA’s CHMP has issued a positive opinion recommending the approval of Opzelura for non-segmental vitiligo in adults & adolescents aged 12yrs. with facial involvement
- The opinion was based on the 2 P-III trials (TRuE-V1 & TRuE-V2) evaluating ruxolitinib cream vs vehicle in 600+ patients. The results showed a significant improvement in facial & total body repigmentation as shown by the no. of patients reaching the F-VASI-T-VASI EPs @24wk. with a higher proportion of patients responding at 52wk.
- Opzelura was approved in the US for the topical treatment of nonsegmental vitiligo & is also approved for the topical short-term & non-continuous chronic treatment of mild to mod. AD in non-immunocompromised patients
PRX–102
Active ingredient: pegunigalsidase alfa Approved: February 25, 2023
Company: Protalix BioTherapeutics Disease: Fabry Disease
- The EMA’s CHMP has adopted a positive opinion recommending marketing authorization for PRX-102 to treat adult patients with Fabry disease. The EC’s final decision on the MAA is expected in May 2023
- The opinion was based on positive data from a comprehensive set of preclinical, clinical & manufacturing studies & clinical programs incl. P-III (BALANCE), (BRIDGE) & (BRIGHT) trials, the P-I/II trial, and ongoing related extension studies. The tolerability & immunogenicity profiles of PRX-102, acc. to data from the clinical program suggested that it has the potential to be a long-lasting therapy
- PRX–102 is a novel recombinant human α–Gal–A enzyme that is being investigated as an enzyme replacement therapy (ERT) for Fabry disease
Libtayo
Active ingredient: cemiplimab Approved: February 27, 2023
Company: Regeneron Disease: Non-Small Cell Lung Cancer
- The EMA’s CHMP has adopted a positive opinion of Libtayo + Pt-based CT for advanced NSCLC with ≥1% PD-L1 expression. The EC’s final decision is expected in the coming months
- The opinion was based on the P-III trail (EMPOWER-Lung 3) evaluating Libtayo + Pt-doublet CT vs Pt-doublet CT alone in 466 patients with LA or metastatic NSCLC, sq. or non-sq. histologies across all PD-L1 expression levels with no ALK, EGFR, or ROS1 aberrations
- The trial showed an improvement in OS in the overall population, m-OS (22 vs 13mos.) with a median follow-up of 16mos., 45% relative reduction in risk of death, and survival benefit was observed with a median duration of follow-up of 28mos., AEs (25%) of patients and permanent discontinuation due to AEs in 5% of patients
11. AbbVie Receives EMA’s CHMP Positive Opinion of Rinvoq (upadacitinib) for Crohn's Disease
Rinvoq
Active ingredient: upadacitinib Approved: February 27, 2023
Company: AbbVie Disease: Crohn's Disease
- The EMA’s CHMP adopted the positive opinion recommending the approval of upadacitinib for moderately to severely active CD
- The opinion was based on 2 induction studies (U-EXCEED & U-EXCEL) & 1 maintenance study (U-ENDURE) evaluating upadacitinib (45mg, qd) as IT & 15/30mg, qd as MT vs PBO. In all P-III studies, patients achieved the co-primary EPs of clinical remission per SF/AP & endoscopic response
- Patients also achieved the 2EPs of endoscopic remission, SES-CD ulcerated surface subscore of 0 @12 & 52wks. in patients with SES-CD ulcerated surface subscore ≥1 at baseline, mucosal healing is connected to improvements seen by endoscopy & absence or disappearance of ulceration. The safety profile was consistent with the known safety profile of upadacitinib
Fintepla
Active ingredient: fenfluramine Approved: February 28, 2023
Company: UCB Disease: Seizures
- Fintepla has been approved in the EU for seizures associated with LGS as an add-on therapy to other anti-epileptic medicines in patients aged ≥2yrs.
- The approval was based on safety & efficacy data from a P-III clinical trial evaluating Fintepla vs PBO in 263 patients which showed a greater reduction in the frequency of drop seizures at a dose of 0.7mg/kg/day while no cases of valvular heart disease or pulmonary arterial hypertension were reported
- Fenfluramine is a serotonin-releasing agent that stimulates multiple 5-HT receptor sub-types through the release of serotonin. To ensure consistent cardiac monitoring & to reduce potential off-label use, fenfluramine oral solution is available through a controlled access program
Note: PRX–102, Akeega, Vafseo, Opzelura, Libtayo & Rinvoq received EMA’s CHMP Positive Opinion & EC’s Conditional Marketing Authorization for Hemgenix, Triumeq PD
Related Post: Insights+: EMA Marketing Authorization of New Drugs in January 2023
Tags

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.














